
University of Geneva - Faculty of Medicine

Others Links
Faculty of Medicine
CMU-Library
Medline-Pubmed
Host Pathogens Interactions
Home
| The Viollier Lab | |||
|---|---|---|---|
|
 |
Pr. P. VIOLLIER CMU/ Dpt MIMOL Rue Michel-Servet 1 1211 Genève 4 Suisse Patrick.Viollier@unige.ch Tel.: 022 379 41 75 Fax: 022 379 57 02 |
|
| Research's subject | Group's publications | Team's members | To Find |
| _________________________________________________________________ | |||
| Research's subject | |||
| Cell cycle control in C.crescentus, an asymmetric bacterium. | |||
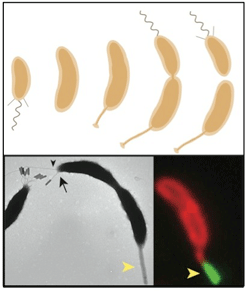 Eukaryotic and prokaryotic cells must execute cellular events at a precise time during cell division cycle. To dissect the molecular mechanisms for this cell cycle coupling, we exploit the powerful forward genetics and the synchronizeability offered by Caulobacter crescentus. The preeminent model system for studies on the cell cycle and differentiation in bacterial cells, Caulobacter differentiates the cell poles at specific stages in the cell cycle before dividing asymmetrically into two functionally specialized daughter cells featuring distinct polar organelles. In past research we uncovered several cell cycle -regulated organizers of polar differentiation that direct organellar components to the proper subcellular assembly site. In other work, we also unmasked a novel regulatory mechanism that links the cell cycle transcriptional program with cytokinesis (cell constriction) in response to a metabolic cue provided by NAD(H). We are currently using genome-wide chromatin-immunoprecipitation techniques (ChIP-SEQ) to decipher the transcriptional program that underlies Caulobacter development with the goal of unearthing uncharacterized cell cycle-regulated genes. We will also determine if and how the transcriptional program of Caulobacter is re-wired upon infection with a bacteriophage or if, in turn, the bacteriophage replication cycle is tuned to that of its host. With this simple bacterial model system we hope to illuminate principles of how viral and host transcriptional programs are coordinated and/or suppressed. Eukaryotic and prokaryotic cells must execute cellular events at a precise time during cell division cycle. To dissect the molecular mechanisms for this cell cycle coupling, we exploit the powerful forward genetics and the synchronizeability offered by Caulobacter crescentus. The preeminent model system for studies on the cell cycle and differentiation in bacterial cells, Caulobacter differentiates the cell poles at specific stages in the cell cycle before dividing asymmetrically into two functionally specialized daughter cells featuring distinct polar organelles. In past research we uncovered several cell cycle -regulated organizers of polar differentiation that direct organellar components to the proper subcellular assembly site. In other work, we also unmasked a novel regulatory mechanism that links the cell cycle transcriptional program with cytokinesis (cell constriction) in response to a metabolic cue provided by NAD(H). We are currently using genome-wide chromatin-immunoprecipitation techniques (ChIP-SEQ) to decipher the transcriptional program that underlies Caulobacter development with the goal of unearthing uncharacterized cell cycle-regulated genes. We will also determine if and how the transcriptional program of Caulobacter is re-wired upon infection with a bacteriophage or if, in turn, the bacteriophage replication cycle is tuned to that of its host. With this simple bacterial model system we hope to illuminate principles of how viral and host transcriptional programs are coordinated and/or suppressed. |
|||